Cutting wasted kits by 9.7% with sample collection redesign
Problem
- Genomics invited 3,000 MassMutual policyholders to a free-benefit genetic test service. 1,620 enrolled over a few months with 1,367 returning a kit. The lab rejected 10% (about 137) for invalid samples.
- Those patients received no result, waited roughly a week for a replacement kit, collected another sample, and posted it back. Support handled reships and explanations.
- Each rejection consumed about $40 in kit cost plus shipping and support time. At scale, repeat kits and delays would erode patient and customer trust and make growth unviable.
What I did
Found root causes with the lab to target fixes
- 5% of kits didn't have the patient's date of birth on the sample device, making them unidentifiable.
- 5% had unusable or missing samples.
- A patient survey showed many found the instructions complex.
"Consider rewriting the instructions, they were tricky" — Patient, September 2022
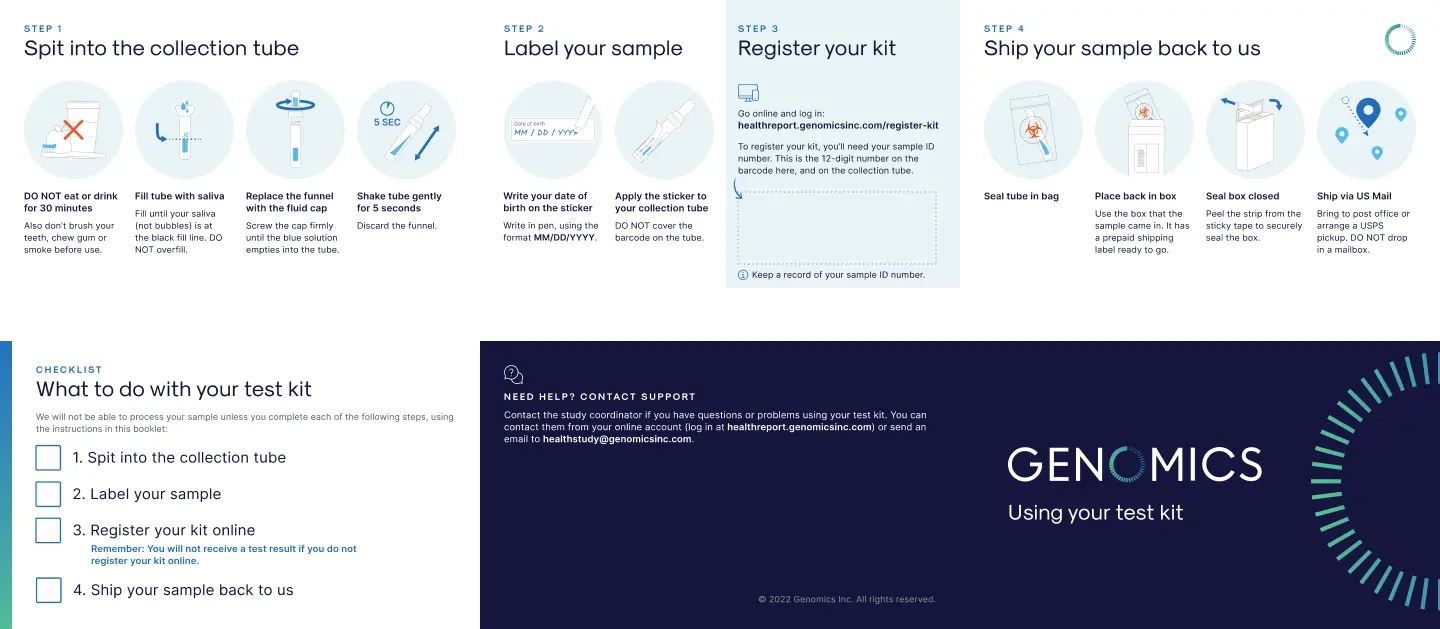
Original paper instructions.
Linked kits to patients with activation codes to stop ID errors
I ran a cross-functional ideation workshop with product, engineering, supply and regulatory teams, settling on a high-effort, high-value solution for maximum return at scale.
- Patients enter a five-digit activation code online instead of writing date of birth on the device. This speeds setup, removes handwriting errors, and enables real-time validation.
- The system validates the code against the database and links the kit to the patient, so the lab identifies the sample on receipt.
- With a valid code, patients follow step-by-step online instructions to collect and return the sample.
- With an invalid code, the system blocks progress and offers fixes. The system requires activation for every kit, addressing the unidentifiable 5% by forcing identification.

Cross-functional ideation workshop.
Redirected patients online via kit design to reduce paper-first mistakes
- I overhauled the kit to align with Genomics' new Health Insights brand identity, meeting patient expectations and fostering trust.
- DNAGenotek supplied the sample device inside the kit which included the paper instructions. Regulations prevented removal.
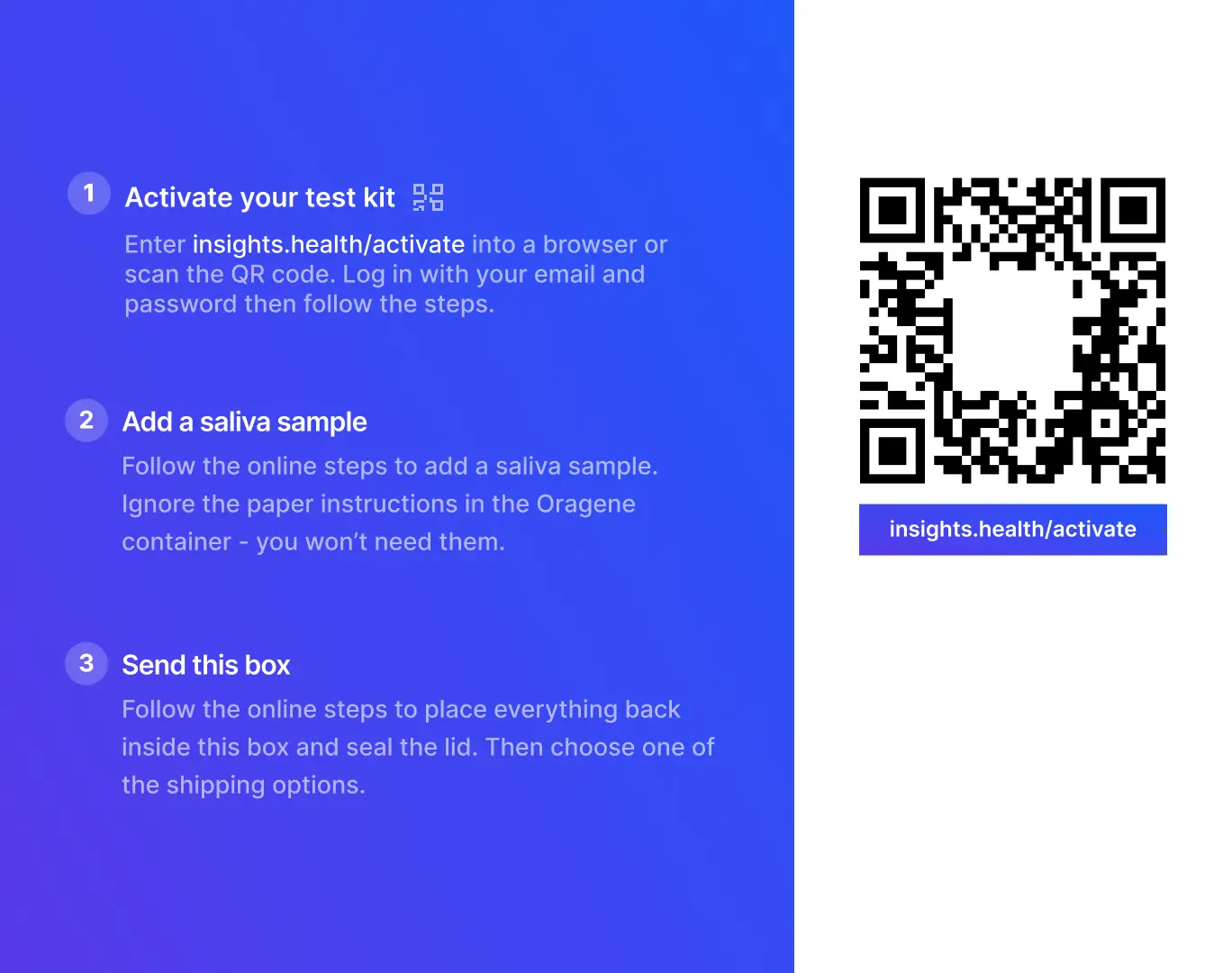
- To mitigate the risk of patients using paper instructions and skipping activation, I added a prominent QR code and additional prompts directing patients online instead.
- Analytics showed 65% of enrolled policyholders were mobile users, so I prioritised a mobile experience while supporting desktop use likely preferred by older patients.

Design exploration for the front of the kit — printed to test the QR and see the print finish quality.

Built step-by-step instructions to lower collection errors
- To address unusable or missing samples, I designed progressively disclosed online instructions with one or two actions per page, using simplified language.
- Instructions responded to browser preferences such as larger text for individual accessibility and were fully keyboard-focusable.
- I included DNAGenotek's illustrations as a quick way to enhance comprehension.
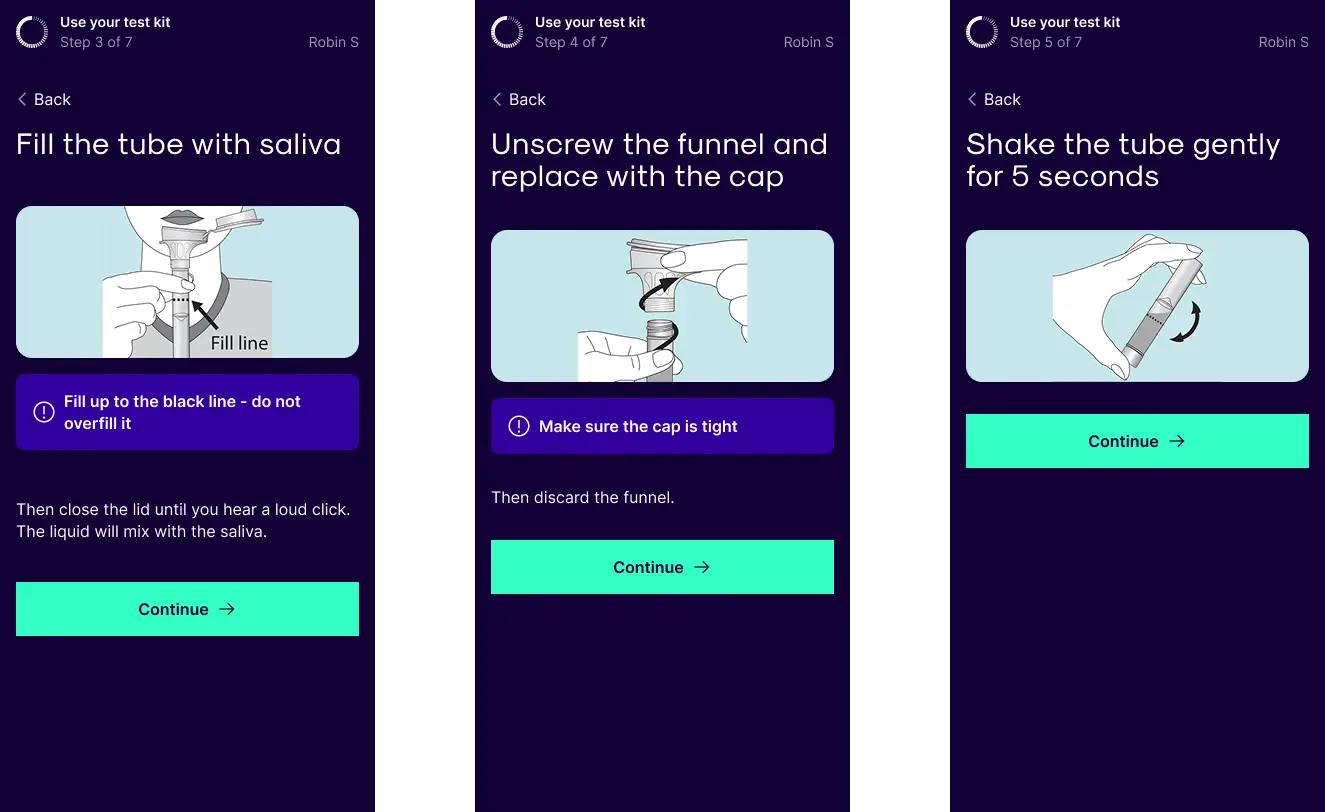
First online instruction prototype. One to two actions per page with simplified copy.
Tested under constraints to surface risks quickly
DNAGenotek couldn't supply extra sample devices in time for US patient testing, so I tested in-person with 5 new UK colleagues unfamiliar with the kit to identify any major issues and maintain momentum. Key kit and instructions findings:
- 3 of 5 read the paper instructions before noticing the QR code.
- Several questioned who DNAGenotek was and raised privacy concerns.
- Some wanted a simple overview of the sampling process upfront.
- Some confused whether a page referred to the kit box or the sample device box.
- All reported the online instructions were easy to use.
Iterated kit and copy to address privacy and box confusion
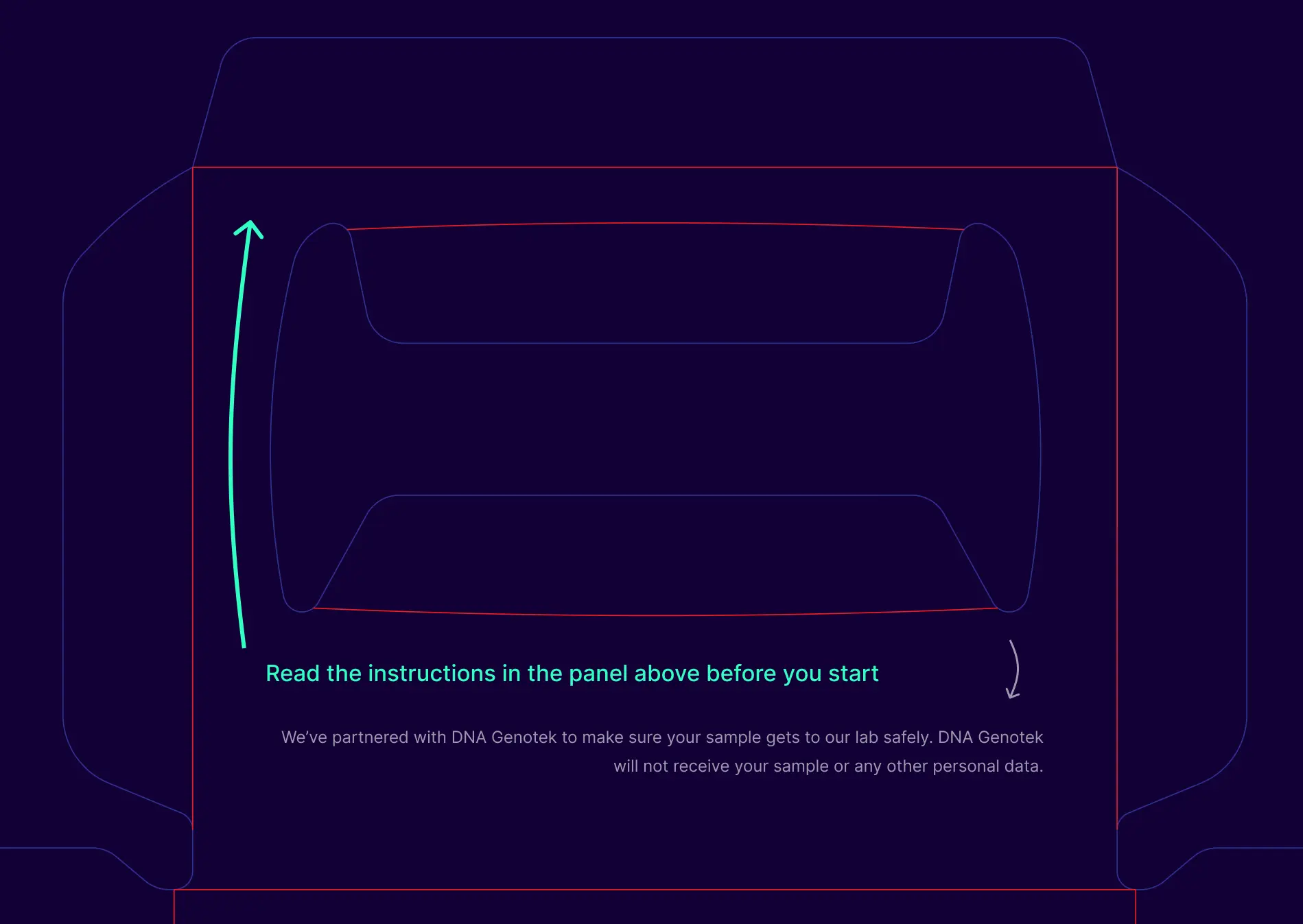
- To address paper-first behaviour and set patient expectations, I pushed the supply chain team to invest in printing on the large inside panel of the kit (about $0.40 or $400 per 1000 kits). I then designed an eye-catching set of steps with the QR alongside.
- With the QR moved to the inside panel, I used the freed up space below the sample device for supporting signposts. To address the partnership with DNAGenotek and relevant privacy concerns, I added content that explained the partnership and stated they do not receive patient personal data or samples both on the kit and in the online journey.
Exploring design options for the inside panel.

Exploring new instructions signpost on the kit print.

Navigated regulatory limits while keeping guidance clear
- To further reduce the risk of paper-first behaviour, in the online instructions I added a first-page callout advising patients to ignore the paper version. Regulatory rules required softer wording to prevent CLIA non-compliance; as a compromise, I asked patients to refer to them only if they need to.
- DNAGenotek's illustrations were off-limits, so I designed custom ones which made the experience more cohesive.
- A later virtual usability test with 5 US colleagues showed no major threats - none referred to the paper instructions and all found the online ones clear.

Illustrations for each step in the sample collection process.
Results
- The reject rate reduced from 10% to 0.3%, saving roughly $3,880 per 1,000 kits in kit costs alone.
- About $2,000 was saved in kit costs in the first month, while reducing delays for patients and protecting trust in the service.
- The improvements reduced reships, shortened time to results, and strengthened confidence in the service.
Key learning
Small usability issues in physical-digital healthcare journeys can create large operational costs. Designing clear guidance and validating identity early prevented downstream failures.
